ENGINEERING
SPECIALIZED ENGINEERING CAPABILITIES ENSURING QUALITY, SAFETY AND SPEED
MARLE GROUP's Engineering Department supervises and streamlines the industrial development process, to accelerate production and thereby improve time to market.
DISTINGUISHED VALUE-ADDED INDUSTRIAL EXPERTISE
- Distinctive skills and experience in orthopedics
- Selection, validation and development of alloys and coatings
- Simulation and design for manufacturability
- Rapid Prototyping (3D printing/silicone tooling/dedicated machining center)

FORGING
WORLD-CLASS QUALITY AND PRODUCTION CAPABILITIES
MARLE GROUP's forging facilities are capable of producing a vast number of implants with superior, consistent physical and mechanical properties.
- Titanium, cobalt-chrome and stainless-steel forging alloys
- High Precision Forging (net shape and near-net shape)
- Control of grain size and orientation to ensure maximum mechanical strength
- Heat-treatments to promote optimal material behavior
- Exceptional dimensional stability
- Stable metallurgical properties


CASTING
STATE-OF-THE-ART INVESTMENT CASTING FOUNDRY ENSURING PRECISION, RELIABILITY AND COST EFFECTIVENESS
MARLE GROUP has industrial casting capacities specifically adapted to the production of orthopedic implants.
- Ample freedom of design, notably for shoulder and knee implants
- Extensive choice of alloys and coatings specific to the medical sector
- Ability to maintain high dimensional accuracy and fine product details
- Highly reproducible and stable manufacturing process
- Specific heat treatments to improve material performance
With two casting production lines, each consisting of an induction furnace and a cluster firing furnace, MARLE GROUP's production facilities can be adapted to accommodate both high and low volume series and a wide variety of product types, offering our customers maximum design flexibility.

MACHINING
ADVANCED MACHINING TECHNOLOGIES FOR COMPLEX IMPLANT DESIGNS
MARLE GROUP has a comprehensive range of 3-, 4-, 5- and 6-axis machining centers that complement our forging and casting technologies, providing great flexibility in terms of design and finishing.
Our expertise in the processes of milling, turning, grinding and electrical discharge machining (EDM) accommodate an extensive range of your orthopedic machining needs.


FINISHING AND POLISHING
COMPREHENSIVE FINISHING TECHNOLOGIES TO MEET THE STRINGENT REQUIREMENTS OF THE ORTHOPEDIC INDUSTRY
MARLE GROUP has developed a comprehensive range of finishing techniques that deliver extremely high-precision finished products, be it for prototypes or high volume series production.
With several robotic polishing units and over 80 highly skilled polishers specialized in orthopedic devices, MARLE GROUP has one of the largest and most experienced polishing teams in Europe, enabling us to deliver a high-quality service, regardless of production volume.
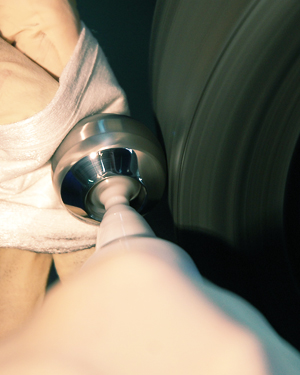

COATING
COATINGS FOR NEXT-GENERATION IMPLANTS
MARLE GROUP's medical coating technologies improve the biomechanical properties of your implants (i.e. higher tensile strength, resistance to corrosion and wear, osseointegration).
Our medical coatings are specifically designed for orthopedic devices, making use of atmospheric plasma spray (APS) and vacuum plasma spray (VPS) technologies. These coatings can be applied to all types of joint prostheses – hip, knee, spine, shoulder, ankle, elbow, hand, foot, osteosynthesis, etc. – thus improving the biological fixation of your implants in cortical and cancellous bone.


CLEANING AND PACKAGING
CLEANED AND PACKAGED IMPLANTS, READY FOR STERILIZATION
MARLE GROUP provides comprehensive cleaning and packaging services in our clean rooms, rendering your implants ready for sterilization.
These capabilities enable us to offer our customers a turnkey service that streamlines your manufacturing process flow while meeting stringent quality requirements.
MARLE GROUP's production lines are equipped with automated chemical or ultrasonic cleaning stations that guarantee maximum microbiological safety for the implants produced, which are therefore directly ready for sterilization.
Packaging operations are carried out in controlled atmospheres or clean rooms:
- 2-ply pouches or double blister packs
- Packaging in cardboard or polypropylene boxes
- Custom packaging


ADDITIVE MANUFACTURING
A SPECIALIST IN ADDITIVE MANUFACTURING OF MEDICAL DEVICES
We provide a wide variety of engineering, R&D and production services for 3D printed medical devices.
MARLE GROUP has an ISO 13485-2016 certified design office that can assist you in the design and development of your medical implants. Using our numerical simulation analysis tools, we can provide biomechanical validation based on the ASME V&V 40 protocol and the requirements of the new Medical Device Regulation (MDR).
Our team of engineers specialized in biomechanics can assist you in developing your orthopedic implant projects and Class I-III medical devices. By combining both a medical and industrial approach, we can optimize and streamline the design of your products with our proficiency in additive manufacturing processes.
The qualified technologies used in our additive manufacturing production center allow us to produce prototypes, patient-specific custom items and series production using both metal and polymer-based technologies.


